Electron Configuration Practice Worksheet Answers. In one area of Australia, the cattle didn’t thrive regardless of the presence of suitable forage. Electrons refill these orbitals in an order and right here is the cheat sheet that helps you better understand the order of orbital diagrams. Write a set of quantum numbers for every of the electrons with an n of three in a Sc atom. This actually isn’t stunning, given the aufbau precept idea that every component may have the electrons in the same place as the component earlier than it, plus one.
Electron Configuration of any factor solely reveals about the electron distribution among atomic orbitals around the nucleus of an atom. Whereas orbital diagram is an illustrative representation of location and spin of the electrons throughout the orbitals within the type of arrows. Read Book Electron Configuration Worksheet Answer Electron Configuration Worksheet Answer Thank you very a lot for reading electron configuration worksheet reply.

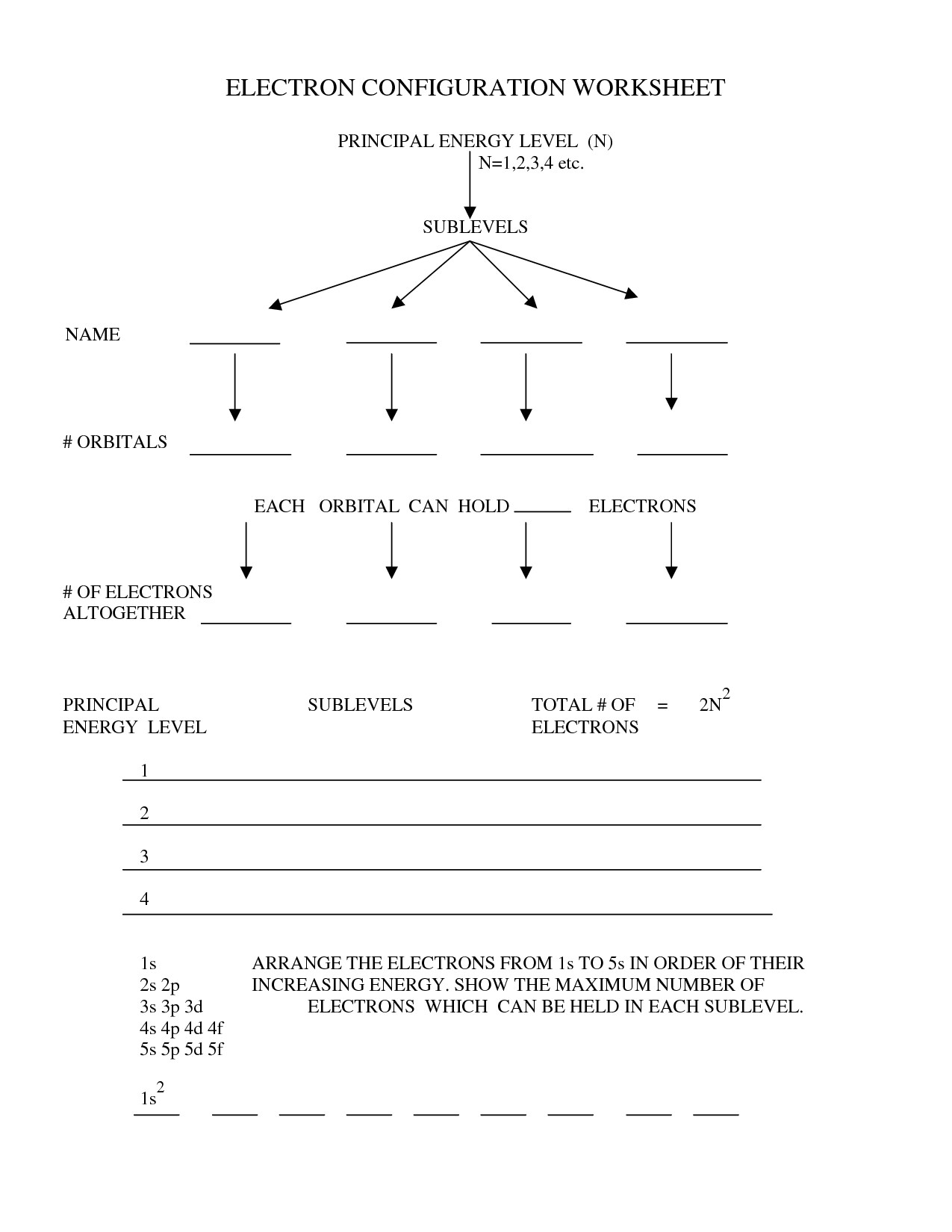
Electron Configurations Video Worksheet. Chemists write electron configurations to explain and communicate the arrangement of electrons around the nucleus of atoms.
Electron Configuration Clarification
Writing digital configurations for the elements current within the preliminary intervals and teams of the periodic desk is simple and easy. But writing digital configuration of elements in the periodic table that come after noble gas group is lengthy and tedious.

4) Make use of periodic table rows and decide orbital shells. Then, determine the valence electrons primarily based on outermost shell electrons and orbital shells.
Definition And Fundamentals Of Electronic Configuration
More follow answer key to write down configuration practices worksheets. 4-5-Practice-Problems-answerspdf.

Electron configuration follow worksheet answer key pogil. Electron configuration practice worksheet reply key chemistry. The follow is an important factor in master any talent.
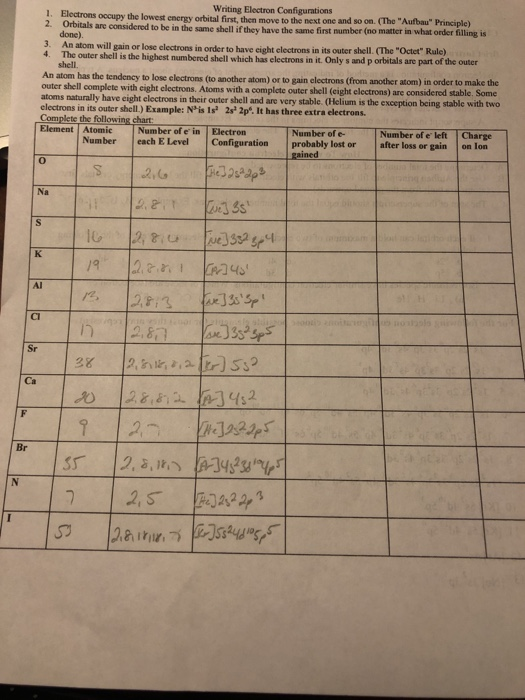
Alexandra Walker Chemistry 3: Electron Configuration Apply Worksheet
This actually isn’t surprising, given the aufbau precept idea that each element may have the electrons in the identical place because the factor before it, plus one. However, it ought to make you kind of marvel why anyone would go to the trouble of writing a bunch of phrases that will all be the same, anyway.
The letters in the electronic configuration of any element i.e. s, p, d, and f symbolize the 4 completely different atomic orbitals. The electron configuration of a component is a standard representation of its electron arrangement within the orbitals of its atom. This notation additionally helps in understanding the bonding capability of electrons in an atom via magnetic and other chemical options.
Fox Eight Randolph County Colleges
The oxygen atom consists of 6 valence electrons and a pair of lone pairs. So, it could bond to central atom utilizing double bond. In chemistry, electron dot configuration has its own significance and this representation of valence electrons was invented by American chemist Gilbert Newton Lewis.

To put it simply, each particular person electron encompasses of 4 quantum numbers and two electrons must exhibit reverse spins when positioned in the identical orbital. Well, positively charged electrons get attracted by negatively charged electrons whereas probably charged electrons repel each other. Click the PDF to check the solutions for Practice Questions.
This notation aids in predicting how atoms will join together to from chemical bonds and their conduct. The more electron configuration apply issues you do the better you’ll. S, P, D and F are the four different atomic orbitals situated across the nucleus of an atom with completely different energy levels.
Or, in different words, it’s written as a substitute of 1s22s22p63s23p64s23d104p65s24d105p6when writing electron configuratons. Different electron configurations may have totally different numbers and letters and such, however they’ll all comply with the same basic sample. For understanding the complete image of atomic spectra of parts in the periodic table.
Abbreviated Electron Configuration
SolutionAlthough each and are appropriate, encompasses each and is the most effective reply. 6) Check out for each atom whether it possess octet configuration.

Answer key Electron Configurations Assignment -online student copy.pdf. Keeping the uncertain behaviour of electrons in mind, our scientists discovered completely different energy ranges across the nucleus of an atom. And additionally said that these atomic orbitals encompasses of electrons at highest possibility.

If you look at these two electron configurations, you can most likely inform the distinction between them after a few minutes of wanting. In lead, the final term is 6p2, whereas in bismuth it’s 6p3. Hence, unabbreviated electron configuration stays for much longer, confused and time-taking.
And whereas replacing the noble gas factor is written in square brackets. In this way, abbreviated electron configuration is much more useful for components that has greater atmic numbers.

What is Hund’s rule of most multiplicity? And that’s the only distinction between the d-orbitals and the other ones.
Electrons refill these orbitals in an order and right here is the cheat sheet that helps you better understand the order of orbital diagrams. History of atomic principle worksheet answer key … Related photographs to tell you more.

What this implies is that we have to add one other term to compensate for the electron within the new location. It allows us to trace where the electrons in a chemical response go.

It helps us to know the reactivity of varied components. It turns out that electron configurations give us info such as how many bonds something will wish to make, in addition to how many electrons one thing needs. That’s useful, provided that chemical reactions are actually simply quests for electrons.

Therefore, we are ready to say that the transcribed description of orbital diagram is nothing however electron configuration. As the name proposes, ‘n’ is the chief power stage where the electron is well detectable. And the ‘n’ value is set based on the gap of energy degree from the nucleus of the atom.

The ‘ℓ’ values remains between zero and n-1 whereas relying on the values of principal quantum quantity. Here, if the n worth is 2, then the ‘ℓ’ value is both zero or 1.
1) Analyse the whole variety of valence electrons of every atom in a molecule. The digit on the ones place of the group number refers again to the variety of valence electrons of a component.

Either means, if you hold studying, you’ll be a professional at electron configurations by the end of this tutorial. Using full subshell notation , predict the electron configurations of the following ions.
These values vary begin from 1 to n…, while n denotes the value of the outermost shell occupied with electron. As all of us already know, electrons bear charge i.e. both negative or optimistic, and are free to vary their places often.

Based on this data, let us learn about floor and excited state levels and in addition concerning the variations between these two states of energy levels. Hund’s rule denotes that electrons should occupy each single orbital of a subshell with at least one electron with similar spin direction.

The electrons which do not participate in any type of chemical bonding and do not refer to valence electrons are core electrons. These electrons are normally present in inner power levels and fully occupied and therefore referred to chemically inert electrons. So with the assistance of orbital diagram, we will easily discover out which sort of atomic orbitals crammed out and that are partially occupied with electrons.

An online noble fuel electron configuration calculator supplies a condensed method of finding the electron configuration, atomic quantity, and atomic mass of given. This line be related to electron configuration worksheet 2 reply key.

Having a Periodic Table obtainable may even make the sample more seen. Use the patterns within the periodic table to write down the longhand electron configuration notation for the following parts.

In this exercise, you’ll first observe a simulated mild spectrum of hydrogen gasoline. Electron configuration follow our primary …

Are electrons going from ground states to excited states? It’s good to know what electrons we’re speaking about in addition to what states they’re transferring from and to.

The ground state electron configuration of Fe is _____. The floor state electron configuration of Ga is _____.
Now, you might recognize this as being completely different than your regular periodic table. The major concept is the same, however you’ll notice that numerous sections have been colored and labeled. These refer to the kinds of electrons that are present in the outer electrons of every element.